Polyprotic Acid Base Equilibria Problems, pH Calculations Given Ka1, Ka2 & Ka3 - Ice Tables - YouTube

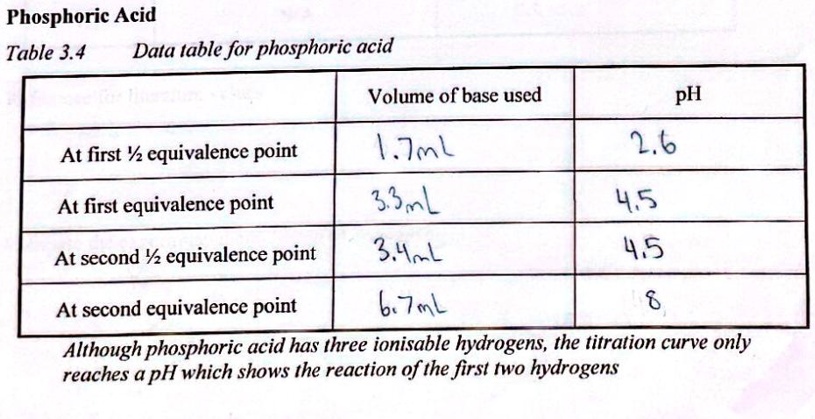
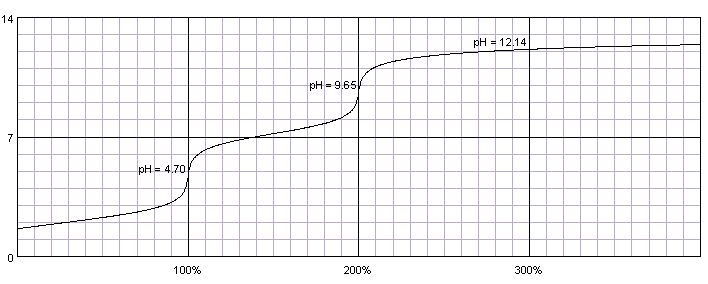
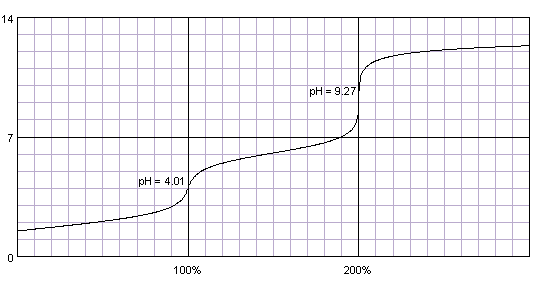
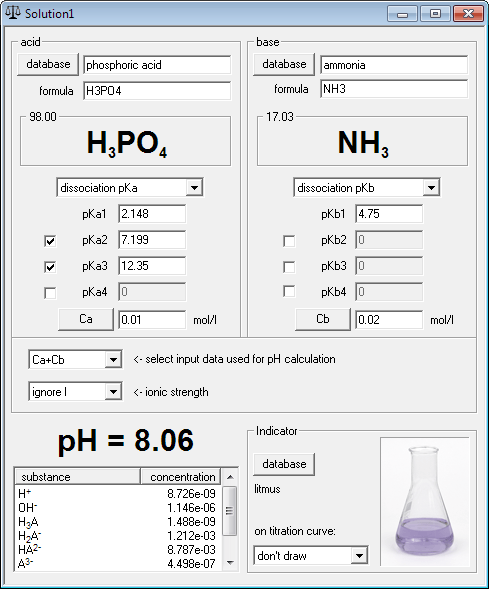
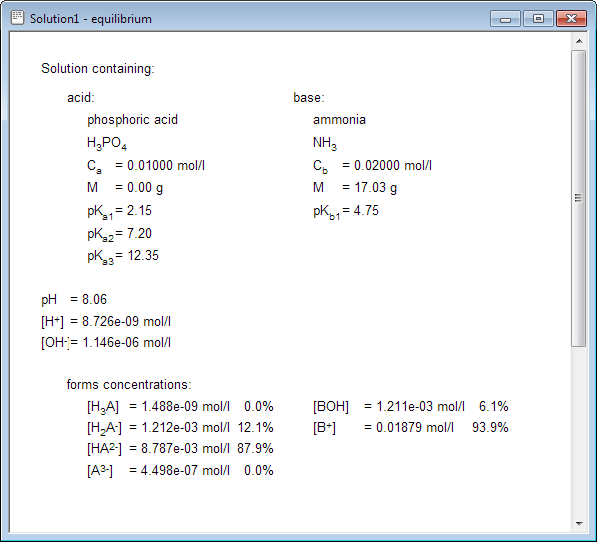
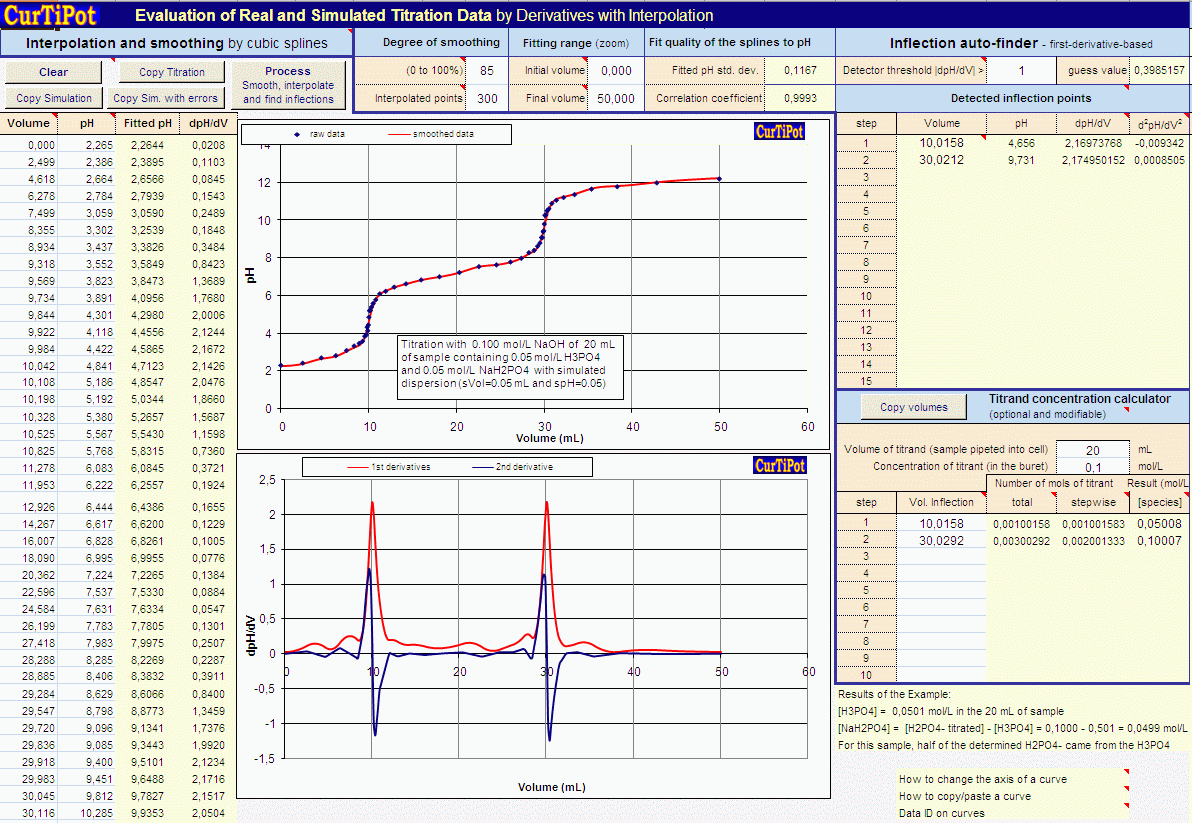
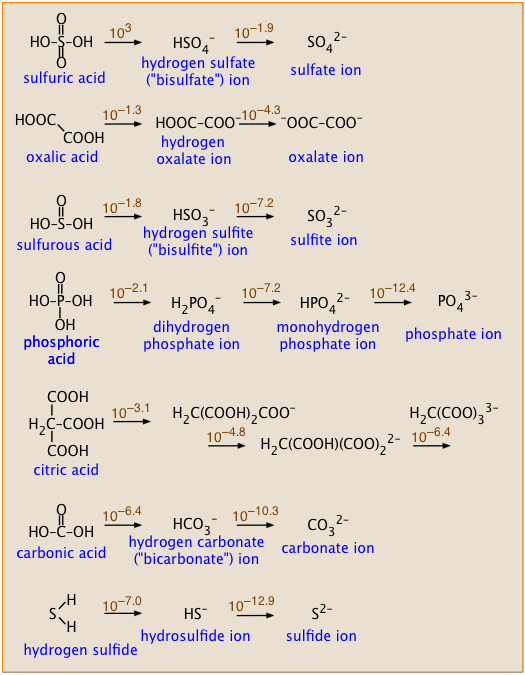
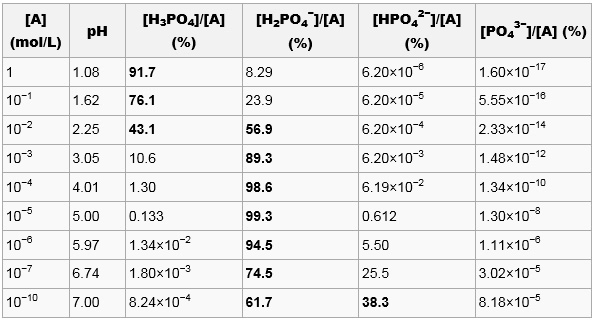
Calculate pH of a 0.1 M Na2HPO4 solution. K1, K2 and K3 for H3PO4 are 7.1 × 10^-3 × 6.3 × 10^-8 and 4.5 × 10^-13 respectively. Which approximation is necessary for the calculation?