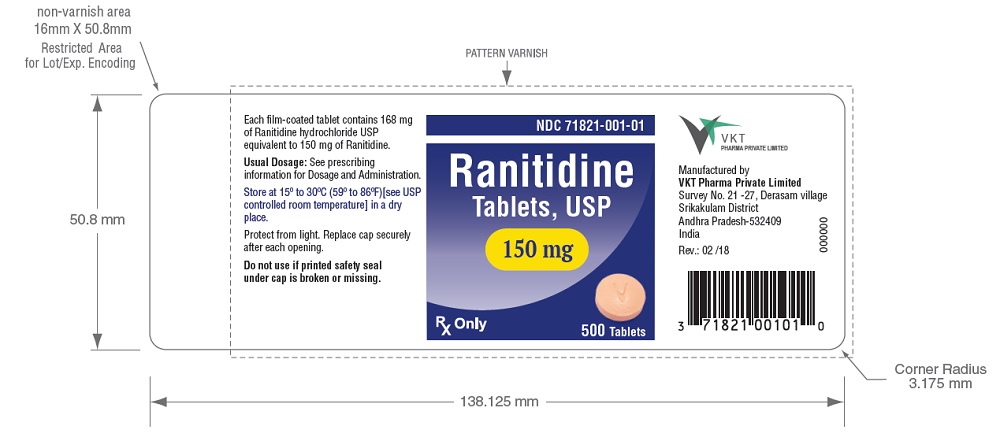
EPINK Health - Recalled ranitidine drugs had excessive amounts of NDMA carcinogen Recalled products containing ranitidine are: 1. Zantac Injection 50mg/2ml (MAL19950591ARZ) 2. Zantac Syrup 150mg/10ml (MAL19910829ACRZ) 3. Zantac Tablet 150mg (not

Healthy Rani-150 Ranitidine Hydrochloride Tablets IP 150 Mg, Healthy Life Pharma, 10 X 10 Tablet at Rs 5.50/strip in Mumbai